 The molecular vibrations are harmonic (at least to good approximation), and the atoms oscillate about their equilibrium positions, even at the absolute zero of temperature. (To some extent rotation influences the geometry via Coriolis forces and centrifugal distortion, but this is negligible for the present discussion.) In addition to translation and rotation, a third type of motion is molecular vibration, which corresponds to internal motions of the atoms such as bond stretching and bond angle variation. The overall (external) quantum mechanical motions translation and rotation hardly change the geometry of the molecule. Since the motions of the atoms in a molecule are determined by quantum mechanics, "motion" must be defined in a quantum mechanical way. The molecular geometry can be described by the positions of these atoms in space, evoking bond lengths of two joined atoms, bond angles of three connected atoms, and torsion angles ( dihedral angles) of three consecutive bonds. The position of each atom is determined by the nature of the chemical bonds by which it is connected to its neighboring atoms. The molecular geometry can be different as a solid, in solution, and as a gas. Geometries can also be computed by ab initio quantum chemistry methods to high accuracy. Larger molecules often exist in multiple stable geometries ( conformational isomerism) that are close in energy on the potential energy surface. Molecular geometries are best determined at low temperature because at higher temperatures the molecular structure is averaged over more accessible geometries (see next section). NMR and FRET methods can be used to determine complementary information including relative distances, Īngles, and connectivity. Gas electron diffraction can be used for small molecules in the gas phase. X-ray crystallography, neutron diffraction and electron diffraction can give molecular structure for crystalline solids based on the distance between nuclei and concentration of electron density. IR, microwave and Raman spectroscopy can give information about the molecule geometry from the details of the vibrational and rotational absorbance detected by these techniques. The molecular geometry can be determined by various spectroscopic methods and diffraction methods. they can be understood as approximately local and hence transferable properties. The angles between bonds that an atom forms depend only weakly on the rest of molecule, i.e. Molecular geometry influences several properties of a substance including its reactivity, polarity, phase of matter, color, magnetism and biological activity. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. The oxygen has two bonding electron pairs (single bond to each H) and two non-bonding pairs giving water a #AX_2E_2# conformation and a bent shape.Study of the 3D shapes of molecules Geometry of the water molecule with values for O-H bond length and for H-O-H bond angle between two bonds #H_2O# we need to consider the central atom of water which is oxygen. 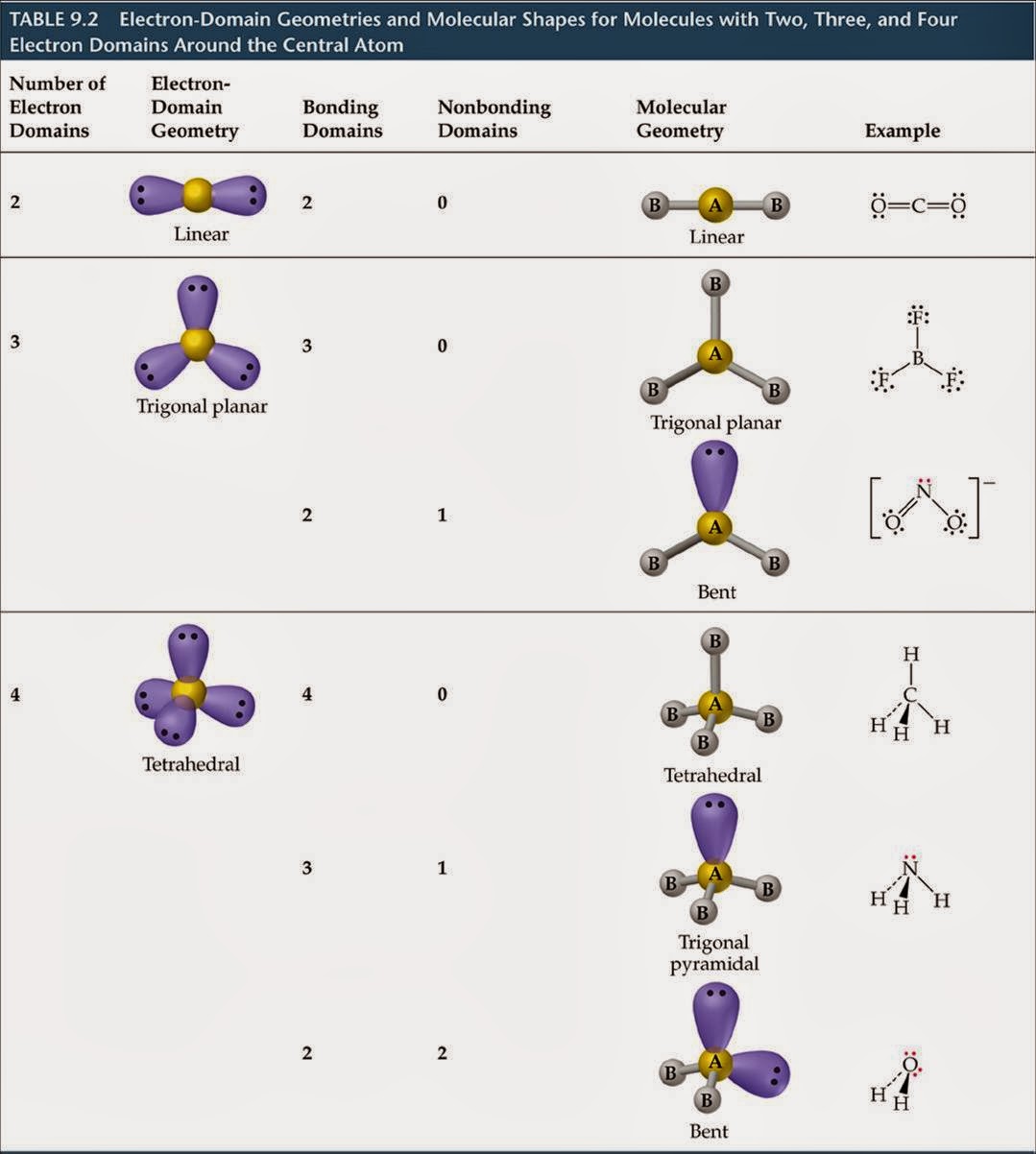
We can use the following notations when examining a Lewis structure of a molecule.Į = non-bonding electron pairs of the central atom This theory basically says that bonding and non-bonding electron pairs of the central atom in a molecule will repel (push away from) each other in three dimensional space and this gives the molecules their shape. VESPR stands for valence shell electron pair repulsion. It applies a theory called VESPR for short. Molecular geometry is a way of describing the shapes of molecules. Similar logic applies to all the shapes, you just have to remember which "spoke" will be taken up by an electron pair. Once there are any electron pairs, one spoke of the original shape gets "eaten up": for example, a #AX_4E_2# is an octahedron shape, but the two "spokes" are taken up by electron pairs, so you're left with just the square-a square planar shape. #6#: octahedron (a flat square with two "spokes") 
#5#: trigonal bipyramid (a trigonal planar shape with two "spokes") #3#: trigonal plane (a flat equilateral-triangle-looking shape) As it has a VSEPR shape #AX_5E_0# it is a trigonal bipyramid.Įach steric number has a same "basic shape": Its steric number is #5# due to the #5# bonded atoms to the central #S# atom plus #0# lone electron pairs. Thus, it is in the form #AX_3E_1#, which forms a trigonal pyramidal shape. 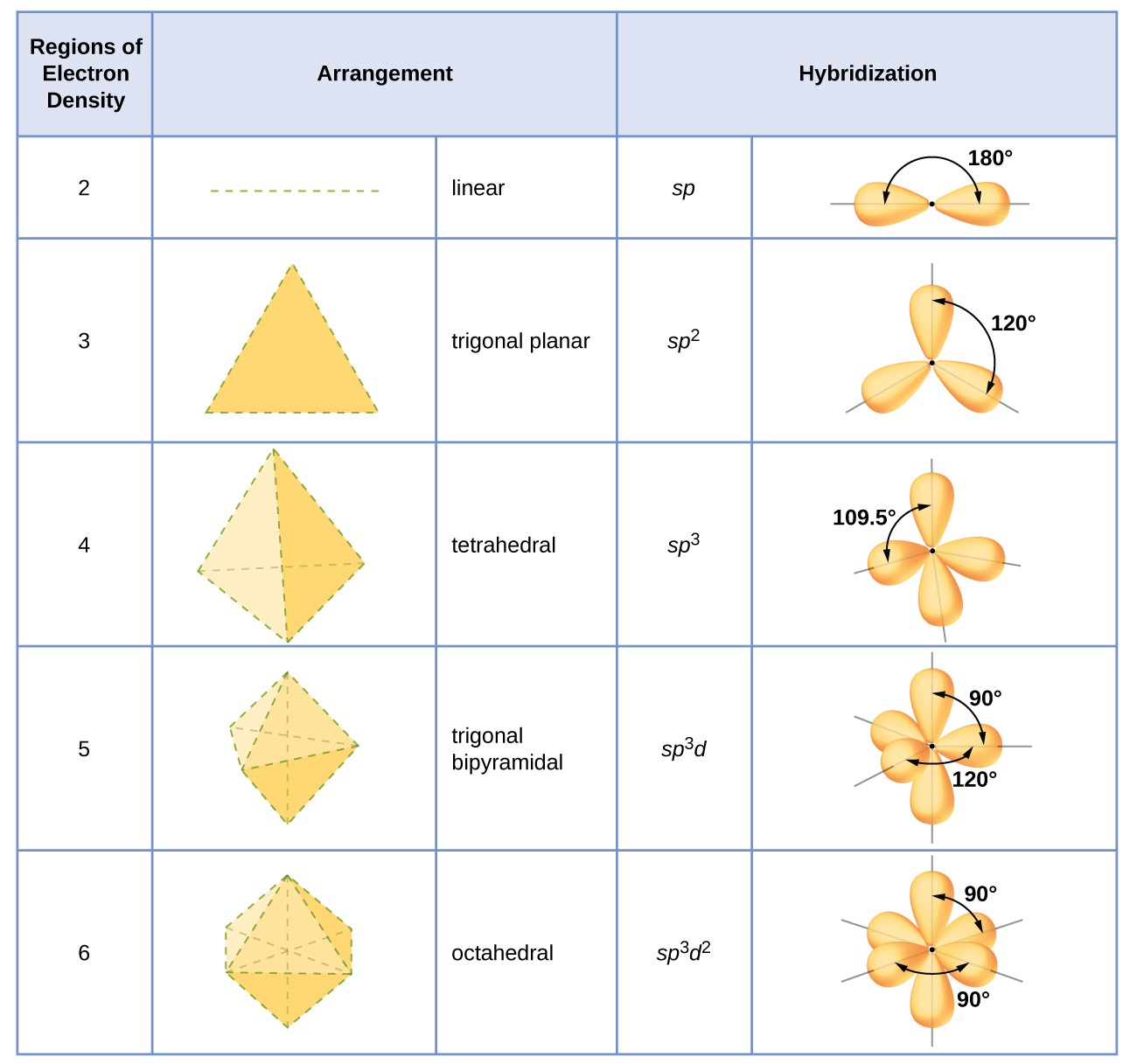
#N#, the central atom, has a steric number of #4#, calculated by the #3# atoms it's bonding with #+1# lone pair. This is the total number of electron pairs and bonds with other atoms. Find the central molecules' steric numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
